 At high pressure, the compressibility factor 'Z' is equal toa
At high pressure, the compressibility factor 'Z' is equal toa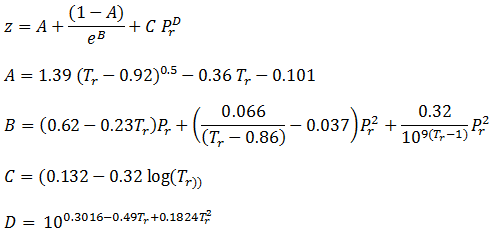 Excel Calculations: Compressibility Factor for Natural Gas
Excel Calculations: Compressibility Factor for Natural Gas Thermodynamics - 3-7 Ideal Gas Equation with compressibility factor example 1
Thermodynamics - 3-7 Ideal Gas Equation with compressibility factor example 1 ChemE 260 Equations of State April 4, 2005 Dr. William Baratuci
ChemE 260 Equations of State April 4, 2005 Dr. William Baratuci Figure 3 from A Simple Equation Of State For Calculating The
Figure 3 from A Simple Equation Of State For Calculating The Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT } (i) What is the value of Z an ideal gas?(ii) For real gas what will be
Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT } (i) What is the value of Z an ideal gas?(ii) For real gas what will be Compression Factor Calculator - Calculator Academy
Compression Factor Calculator - Calculator Academy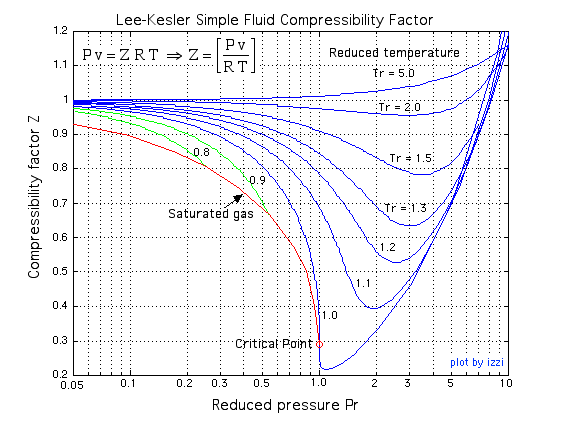 3.3: Real gas and compressibility factor - Engineering LibreTexts
3.3: Real gas and compressibility factor - Engineering LibreTexts If Z is a compressibility factor, van der Waals equation at low
If Z is a compressibility factor, van der Waals equation at low At high pressure, the compressibility factor 'Z' is equal toa)unityb) c) d)ZeroCorrect answer is option 'C'. Can you explain this answer? - EduRev NEET Question
At high pressure, the compressibility factor 'Z' is equal toa)unityb) c) d)ZeroCorrect answer is option 'C'. Can you explain this answer? - EduRev NEET Question Compressibility factor - Wikipedia
Compressibility factor - Wikipedia- Thermo] Derivation of compressibility factor vs reduced pressure
 Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT
Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT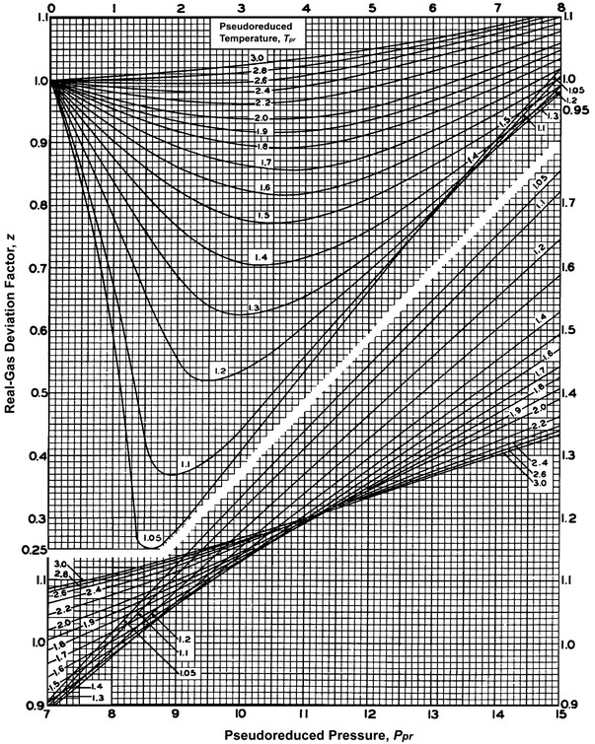 3.3.3: Natural Gas Properties PNG 301: Introduction to Petroleum and Natural Gas Engineering
3.3.3: Natural Gas Properties PNG 301: Introduction to Petroleum and Natural Gas Engineering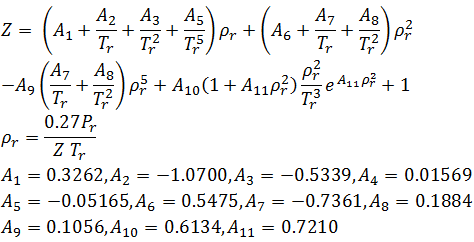 Excel Calculations: Compressibility Factor Calculator for Excel
Excel Calculations: Compressibility Factor Calculator for Excel ChemE 260 Equations of State April 4, 2005 Dr. William Baratuci Senior Lecturer Chemical Engineering Department University of Washington TCD 2: E & F CB. - ppt download
ChemE 260 Equations of State April 4, 2005 Dr. William Baratuci Senior Lecturer Chemical Engineering Department University of Washington TCD 2: E & F CB. - ppt download 3.2 Real gas and compressibility factor – Introduction to
3.2 Real gas and compressibility factor – Introduction to Compressibility factor (gases) - Knowino
Compressibility factor (gases) - Knowino Compressibility Factor from Redlick-Kwong Equations
Compressibility Factor from Redlick-Kwong Equations Figure 3 from A Simple Equation Of State For Calculating The Compressibility Factor Of Pure Fluids Based On The Virial EOS
Figure 3 from A Simple Equation Of State For Calculating The Compressibility Factor Of Pure Fluids Based On The Virial EOS Thermodynamic Properties Property Table w Property Table -- from
Thermodynamic Properties Property Table w Property Table -- from Real gasses For an ideal gas, the compressibility factor Z = PV/nRT is equal to unity for all conditions. For a real gas, Z can be expressed as a function. - ppt
Real gasses For an ideal gas, the compressibility factor Z = PV/nRT is equal to unity for all conditions. For a real gas, Z can be expressed as a function. - ppt- The compressibility factor for a real gas at high pressure is (a) 1+RT/pb (b) 1 (c) 1+pb/RT (d) 1-pb/RT - Sarthaks eConnect
 Real gasses For an ideal gas, the compressibility factor Z = PV
Real gasses For an ideal gas, the compressibility factor Z = PV